Layered double hydroxides (LDHs) are lamellar ionic materials built by stacking positively charged mixed-metal hydroxide layers weakly bound to each other through Van der Waals interactions. Different kinds of guest anions, together with water molecules, occupy the interlamellar space to balance the excess positive charge of the hydroxide sheets, giving a layered hydrotalcite-like structure. The noteworthy capacity to host and exchange different organic or inorganic anions in the interlamellar space has attracted much interest in view of a number of possible applications, such as superhydrophobic surfaces, anti-corrosion coatings, flame retardants, clay-modified electrodes for electroanalytical applications, adsorbents and biosensors for water treatment and purification, nanosystems for molecular and gene storage and drug delivery.
The LDH anion conductivity can be modulated incorporating different various anions in the interlayer, allowing their use as solid electrolytes or as fillers in different electrochemical applications, such as fuel cells, sensors, electrocatalysts and supercapacitors.
In the LIME lab, LDHs were prepared as thin film by a hydrothermal process on metal surfaces, especially Al, that acts as both reactant and sacrificial support. This strategy uses a simple one-step, room-temperature, environmental-friendly method to achieve the controlled growth of well adherent LDH films on metal or metal-coated substrates. The ionic conductivity was characterized as a function of the growth time and for three intercalated anions: NO3–, Cl– and F– with F– ions showing the higher highest ionic conductivity.
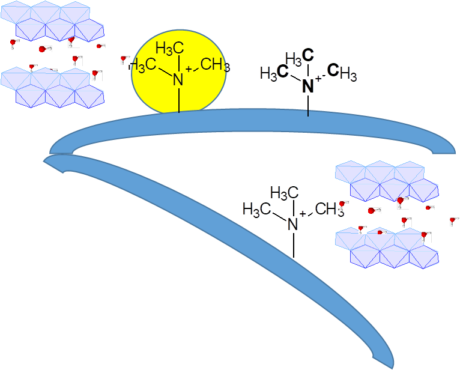
Nanometric MgAl-LDH and ZnAl-LDH were also prepared by co-precipitation methods. This strategy enabled the intercalation of the ionic liquids into the interlamellar space of LDH with the aim of increasing the ionic conductivity and reducing its dependence on the water content. The ionic conductivity in fully humidified conditions reaches reached elevated values, up to 39 mS/cm, a very high conductivity for a hybrid inorganic solid.
LDHs were used as fillers in anion exchange membranes with the aim to mitigate their degradation in alkaline conditions due to the composite effect and reduce the membrane swelling and excessive water uptake. We studied the use of MgAl and ZnAl-LDH as second phase in composites of PSU and PPO with grafted quaternary ammonium groups.